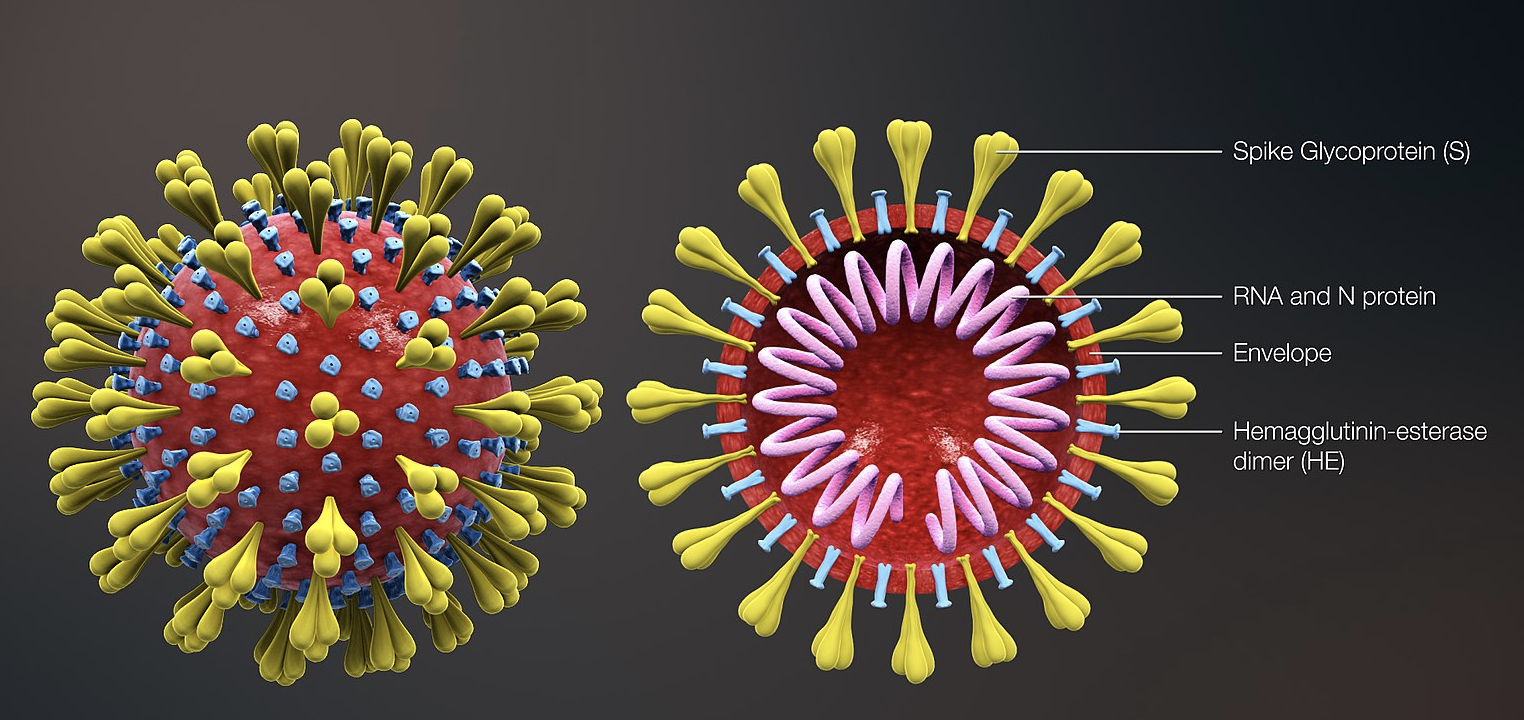
A Phase 1 clinical trial is evaluating an investigational vaccine designed to protect against coronavirus disease in Seattle.
The open-label trial will enroll 45 healthy adult volunteers ages 18 to 55 years over approximately 6 weeks. The first participant received the investigational vaccineon Monday, March 16th.
The study is evaluating different doses of the experimental vaccine for safety, and its ability to induce an immune response in participants. This is the first of multiple steps in the clinical trial process for evaluating the potential benefit of the vaccine.
The investigational vaccine was developed using a genetic platform called mRNA. The investigational vaccine directs the body’s cells to express a virus protein that it is hoped will elicit a robust immune response.
The mRNA-1273 vaccine has shown promising results in animal models, and this is the first tiem it will be used on humans.